Are Cows Getting a Free Pass on Methane Emissions?
January 20, 2025 § Leave a comment
A recent study reported in the Proceedings of the National Academy of Sciences1 convincingly shows that atmospheric methane increases in the last 15 years can be attributed primarily to microbial sources. These comprise ruminants (cows in the main), landfills and wetlands. Yet, policy action on methane curbing has largely been focused on leakage in the natural gas infrastructure. In the US as well as in Canada, policies have fallen short of comprehensive action in the agricultural sector2,3.
Before we discuss the study, first some (hopefully not too nerdy) basics. Methane has the chemical formula CH4. The common variety designated 12CH4, has 6 protons and 6 neutrons in the nucleus. An isotope, 13CH4, has an additional neutron. The 13CH4 to 12CH4 ratio is used to detect the origin of the CH4. The actual ratio is compared to that of a marine carbonate, an established standard. The comparison is expressed as δ13CCH4 with units ‰ and is always a negative number because all known species have a lower figure than that of the standard carbonate. Microbially sourced methane will have ratios of approximately -90 ‰ to -55 ‰ and methane from natural gas will be in the range -55 ‰ to -35 ‰. This is the measure used to deduce the source of methane in the atmosphere.
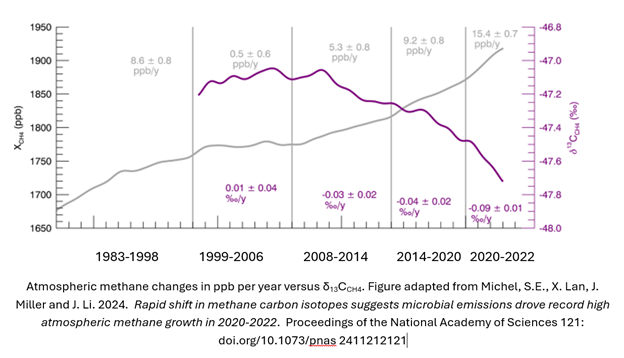
Here is the lightly adapted main figure from the cited study. The data are primarily from The National Oceanic and Atmospheric Administration’s Global Monitoring Laboratory, but as shown in the paper, similar results have been observed from other international sources. The gray line is the atmospheric methane, shown as increasing steadily over decades, but with steeper slopes in the near years. The steeper portion is roughly consistent with the period in which the isotopic ratio becomes increasingly negative. This implies more negative contribution, which in turn means that the main contributory species is microbial. Note also the increased severity of the trend in 2020-2022, and coincidentally or not, increased methane in atmosphere slope in those years. The paper authors do not see the correlation as coincidental. They emphatically state: our model still suggests the post-2020 CH4 growth is almost entirely driven by increased microbial emissions.
A quick segue into why methane matters. The global warming potential of methane is 84 times that of CO2 when measured over 20 years, and 28 times when measured over 100 years. Climatologists generally prefer to use the 100-year figure (and I used to as well), but urgency of action dictates that the 20-year figure be used. The reason for the difference is that methane breaks down gradually to CO2 and water, so it is more potent in the early years.
These research findings point to the need for policy to urgently address microbial methane production. This does not mean that we let up on preventing natural gas leakage, the means to do which are well understood. The costs are also well known and, in many cases, simply better practice achieves the result. In fact, the current shift to microbial methane being a relatively larger component could well be in response to actions being taken today to limit the other source. But it does mean that federal actions must target microbial sources more overtly than in the past. We will touch on a few of the areas and what may be done.
Landfill gas can be captured and treated. In the US, natural gas prices may be too low to profitably clean landfill methane sufficiently to be put on a pipeline. Part of the problem is that, due to impurities such as CO2, landfill methane has relatively low calorific value, almost always well short of the 1 million BTU per thousand cubic feet standard for pipelines. However, technologies such as that of M2X can “reform” this gas to synthesis gas, and thence to methanol, and a small amount of CO2 is even tolerated (Disclosure: I advise M2X).
Methane from ruminants (animals with four-compartment stomachs tailored to digest grassy materials) is a more difficult problem. Capture would be operationally difficult. The approach being followed by some is to add an ingredient to the feed to minimize methane production. Hoofprint Biome, a spinout from North Carolina State University, introduces a yeast probiotic to carry enzymes into the rumen to modify the microbial breakdown of the cellulose with minimal methane production. I would expect this more efficient animal to be healthier and more productive (milk or meat). Nailing down of these economic benefits could be key to scaling, especially for dairies, which are challenged to be profitable. Net-zero dairies could be in our future.
Early-stage technologies already exist to capture methane from the excrement from farm animals such as pigs. These too could take approaches similar to those proposed for landfill gas, although the chemistry would be somewhat different. Several startups are targeting hydrogen production from pyrolysis of methane to hydrogen and carbon. The latter has potentially significant value as carbon black, for various applications such as filler in tires, and biochar as an agricultural supplement. If the methane is from a source such as this, the hydrogen would be considered green in some jurisdictions.
The federal government ought to make it a priority to accelerate scaling of technologies that prevent release of microbial methane into the atmosphere. With early assists, many approaches ought to be profitable. Then it would be a bipartisan play*.
Vikram Rao
*Come together, right now, from Come Together, by The Beatles, 1969, written by Lennon-McCartney
1 Michel, S.E., X. Lan, J. Miller and J. Li. 2024. Rapid shift in methane carbon isotopes suggests microbial emissions drove record high atmospheric methane growth in 2020-2022. Proceedings of the National Academy of Sciences 121: doi.org/10.1073/pnas 2411212121
2 Patricia Fisher https://fordschool.umich.edu/sites/default/files/2022-04/NACP_Fisher_final.pdf
3 Ben Lilliston 2022 https://www.iatp.org/meeting-methane-pledge-us-can-do-more-agriculture
THE FRONT OF THE BOX
December 20, 2011 § Leave a comment
A recent NY Times story has a very interesting take on the environmental movement and changes therein. These organizations in the past have taken national or even global approaches to the issues. The rise of global ambient temperatures caused by greenhouse gases is a case in point.
The general public can be left cold at two levels. One is that global issues do not resonate with a lot of folks, local ones do. The other is the discounting of future privation. This is not unlike discounting future earnings in finance; a discount rate is applied which gives a lower present value. Similarly, future suffering is discounted, especially when it is 40 years out, as are most global warming warnings. Rising water levels on a Florida beach 40 years hence (and only a maybe at that) has little resonance with the public in Wyoming. One could call it two degrees of separation.
The Times story draws a clever analogy. If a consumer is walking down a grocery store aisle and she sees a box with a delectable brownie on the face, she may be attracted to it. Some might look at the back of the box detailing the information indicative of an obese future for the consumer of the goods. Even though the future in this case is more in the short term than the aforementioned global warming one, the choice of looking at the back is personal and will not happen all the time.
 Environmental organizations are credited with focusing simply on the back of the box. This stuff is bad for you, we want saturated fat detailed, and we want the warnings to be explicit, and so on. Interestingly the smoking hazard warnings are in front of the box and likely work better. In this example, the context is local, so that problem is not there. You simply may not get the attention of the consumer.
Environmental organizations are credited with focusing simply on the back of the box. This stuff is bad for you, we want saturated fat detailed, and we want the warnings to be explicit, and so on. Interestingly the smoking hazard warnings are in front of the box and likely work better. In this example, the context is local, so that problem is not there. You simply may not get the attention of the consumer.
According to the story, some of these organizations are getting the message. They are going local and in front of the box. The first is simply a matter of organization, but the second is a bit harder, because the messaging has to hit at the value system. Ocean rise 40 years hence will not play. Asthma risk now for their children will. So, the Sierra Club is focusing on individual coal burning power plants and their presumed effects upon the local population. Shutting these older plants down one by one is the strategy. They have had considerable success and operate in 46 states.
About 40% of coal plants not expected to conform to upcoming EPA standards are over 50 years old. If the Sierra Club and others have their way, it will not matter whether the EPA rules come down. The arguments in Congress over this could put off that day. But if the ill effects of the polluting plants are placed in front of the box for the public, the plants will likely get shut. Thirteen such are currently slated for this fate by Progress Energy in North Carolina.  If shut down effectively through local action, the electricity will still have to be generated in some way. Natural gas is the only viable short to medium term option. The carbon emissions are about half that of coal, and the front of the box arguments regarding particulate emissions, mercury and NOx attributed to coal do not apply. The other option, that of a newer and cleaner coal plant, is not economically justifiable if gas remains relatively cheap. Plentiful shale gas will assure that.
If shut down effectively through local action, the electricity will still have to be generated in some way. Natural gas is the only viable short to medium term option. The carbon emissions are about half that of coal, and the front of the box arguments regarding particulate emissions, mercury and NOx attributed to coal do not apply. The other option, that of a newer and cleaner coal plant, is not economically justifiable if gas remains relatively cheap. Plentiful shale gas will assure that.
However, shale gas is the target of many activists who are fundamentally opposed to all fossil fuel. The back of the box issues of fugitive emissions of methane will not get much traction, especially because of the esoteric arguments involved in the modeling. So they have taken to the matter of methane contamination of water wells, with the powerful backing of a couple of Duke University professors. This is not ideal front of the box material because methane in drinking water is not believed to be a health hazard. But any perceived taint to drinking water is powerful stuff.
The unfortunate aspect to all of this is that it distracts from the real issues, which are use of fresh water and most importantly, the potential for polluting discharge of flow back water from fracturing operations. The methane contamination of water wells, while possible, is easily correctable by best practices, voluntary or forced by rules and penalties. The other two issues require more effort, technical and organizational, and should be the focus of local community action. In the end the combination of effective legislation, technology, and industry cooperation can deliver cheap gas in an environmentally secure fashion. We just need to take the steps to make that happen. Then the side of the box will not matter.
Thanks to Christa WagnerVinson for bringing the NY Times story to my attention
So, Where Did All This Gas Come From Suddenly?
November 13, 2011 § Leave a comment
Few will dispute that shale gas has changed the very make up of the petroleum industry. At every twist and turn new resource estimates appear, each vastly greater than the previous. The estimate in 2008 exceeded the one from 2006 by 38%. As with all resource estimates, be they for rare earth metals or gas, disputes abound. But through all the murk is the inescapable fact: there certainly is a lot of the stuff. How could this suddenly be so? The last such momentous fossil fuel find in North America was the discovery of Alaskan oil. But a discovery out in the nether regions is understandable. In this case we were asked to believe that all this was happening literally in our backyard.
To appreciate what happened we first need to understand how oil and gas is formed and recovered. Millions of years ago marine organisms perished in layers of sediment comprising largely silt and clay. Over time additional layers were deposited and the organic matter comprising the animals and vegetation was subjected to heat and pressure. This converted the matter into immature oil known as kerogen. Further burial continued the transformation to oil and the most mature final form would be methane. By and large the only real difference between oil and gas is the size of the molecule. Methane is the smallest with just one carbon atom. One of the lightest oil components, gasoline, averages about eight carbon atoms. Diesel averages about twelve. So, although we refer to them as oil and gas, chemically they are part of a continuum. So, it is easy to understand that they could come from a single source.
The key word is source. The rock in which the oil or gas originally formed is known as source rock. The figure shows a schematic representation of the location of one such source rock. This is almost always shale, which we told you was some mixture of silt and clay and sometimes some carbonates. Conventionally, the fluid in this rock will migrate to a more porous body.
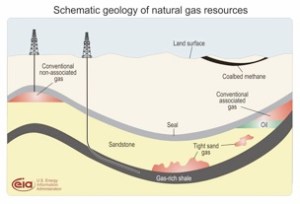
This is depicted as the sandstone shown, which is predominantly silica, an oxide of silicon. It may also be a carbonate, predominantly calcium carbonate. These two minerals are host to just about every conventional reservoir fluid in the world. The fluid (and by the way gas is a fluid, although not a liquid) migrates “updip” as shown to the upper right. This is because the hydrocarbon is less dense than the water saturated rock and essentially floats up, not unlike oily sheens on your cup of coffee.* This migration continues until stopped by a layer of rock through which fluid does not easily permeate. This is known as a seal, and more colloquially, a cap rock. Ironically this is most usually a shale, not unlike where the fluid originated. The trapped fluid is then tapped for production.
The trap is often a dome as shown in the upper left. It can also be a fault. This is when earth movements cause a portion of the formation to break away and either rise or fall relative to the mating part it just separated from. In some instances a porous fluid filled rock will now butt up against an impermeable one, and a seal is formed laterally.

Source: Wikipedia
In the schematic shown the yellow zone would be the sandstone, and the updip fluid shown in red now finds itself abutting an impermeable zone shown in green.
In the early days of prospecting they looked for surface topography indicative of a dome type trap below. These days sound waves reflected back produce excellent images of the subsurface.
Unconventional Gas: We have described how conventional gas, and oil for that matter, are found and produced. The current flurry of activity in shale gas is concerned with going directly to the source. This was previously considered impractical, primarily because the rock has very poor permeability, which is the ease with which fluid will flow in the rock. The permeability of shale is about a million times worse than conventional gas reservoir rock. In fact, as we observed earlier, shale acts as a seal for conventional reservoirs. The breakthrough was the use of hydraulic fracturing. Water is pumped at high pressures, causing a system of fractures. These are then propped open with some ceramic material to hold the cracks open. Without this the sheer weight of the thousands of feet of rock above would close the cracks. The propped open fractures now comprise a network of artificially induced permeability, allowing the gas to be produced. This is akin to pillars and beams used in underground mines.
The sheer ability to extract gas from source rock is now well understood as feasible. But some still doubt the magnitude of the estimated resource. Here is the explanation of why one would expect this resource to be plentiful. Consider that for a conventional reservoir to be formed one needed a confluence of two events. First there needed to be a proximal porous and permeable rock and second, a trap mechanism had to exist. So it would be easy to believe that more source rock did not have these conditions than did. In other words the probability of source rock without a release mechanism was greater than with. This is why it is reasonable to conjecture that the total resource trapped in source rock is greater than the resource that escaped into permeable trapped rock. Further adding to the potential is that this is fresh territory, relatively unexploited. Decades of exploitation have denuded conventional reserves, while the source rock remains relatively untapped.
A word on the nomenclature of resource estimation. A resource estimate indicates the quantity of estimated hydrocarbon accumulation, whether economically recoverable or not. A subset of that is a reserves estimate. Reserves are the portion of the resource that one could recover economically and bring to market. Typically in a new play one would expect reserves to keep getting revised upwards. This is because every new well put on production increases the certainty of the extent and quality of the reservoir, and the reserves can confidently be increased. In reading the popular literature it would be well to keep the distinctions in mind; they are often confused.
*Darker roasts produce more oil. One way to minimize oily sheen is to brew with cold water; also results in a “sweeter” coffee. This is analogous to “sun tea”.
BEYOND GASLAND
October 8, 2011 § 3 Comments
No shale gas production issue may be more fraught with partisan rhetoric than that of water well contamination. The award winning documentary Gasland leveled accusations and energized entire communities. Industry reponse was equally summary in denial. We need to get beyond all that. Here is an attempt at clarity.
Well water contamination is very personal and frightening. Think Erin Brockovich. Airborne species appear not to get the same reaction. Certainly, carbon dioxide in the air barely registers on the average personal anxiety scale. Consequently, assaults on the quality of well water make for avid reading and activism. In the case of shale gas, industry response has also been sweeping in denial. Both sides play fast and loose with the English language, as will be shown.
There are two potential ways in which shale gas operations could contaminate aquifers. One is through leakage of the chemicals used in fracturing. These then would be liquid contaminants. The second is the infiltration of aquifers by produced methane. This is a gaseous contaminant, albeit in the main dissolved in the water. If present, a portion may be released as a gas, as spectacularly depicted in Gasland. Natural occurrences such as the Eternal Flame Waterfall in the Shale Creek Preserve in New York, shown in the picture, demonstrate methane intrusion into a fresh water source.
Natural contamination is either from relatively shallow biogenic methane from decomposing vegetation or from thermogenic gas from deep deposits escaping up along faults and fissures. The last is generally due to tectonic activity at some time. The two types of gas have fairly different fingerprints and can often be distinguished on that basis. Good oil and gas exploitation practitioners will avoid producing in areas with significant vertical leak paths because they vitiate normal sealing mechanisms.
The distinction between potential liquid and gaseous contamination is important because the hazards are different, as are the remedies and safeguards. Also, because well water could not naturally have the liquid contaminants, any presence at all is evidence of a man made source. Therefore, simple testing of wells proximal to drilling operations is sufficient, with the only possible complication being some source other than drilling, such as agricultural runoff. This is easily resolved because of the specificity in the chemicals used for fracturing.
Unfortunately, the two get lumped together in the statements by shale gas opponents and also the genuinely concerned public. Some see methane intrusion as proof of well leakage as a whole and therefore equate it to chemical contamination as well. Gasland reports “thousands of toxic chemicals” as the hazard. In actuality, the mechanisms for possible leakage are quite different. Methane as gas is much more likely to leak out of a badly constructed well than is a liquid. Also, the mechanism by which methane could leak is well understood and it is not conducive to leakage of fracturing fluid. The public cannot be expected to know this and so it is easy to see why the two get banded together. To them, a leaky well is a leaky well. Fortunately, this is not the case.
So, do producing gas wells sometimes leak into fresh water aquifers? The answer is yes. In all cases this is because of some combination of not locating cement in the right places and of a poor cement job. Many wells will have intervals above the producing zone that are charged with gas, usually small quantities in coal bodies and the like. If these are not sealed off with cement, some gas will intrude into the well bore. This will still be contained unless the cement up near the fresh water aquifers has poor integrity. In that case the gas will leak. You will notice nothing in the prior discussion says anything about fracking. In other words a badly constructed well is just that, no matter how the gas was released from the formation.
This distinction is lost on many. The recent notable paper by Robert Jackson and others is the most comprehensive work of its kind to date. It unequivocally shows no fracture chemical intrusion into water wells. It also shows gas intrusion in disturbingly many cases, although later studies will take care to normalize for possible natural seeps and prior drilling activity. Yet the title of the paper is Methane contamination of drinking water accompanying gas-well drilling and hydraulic fracturing. (Emphasis added) The last three words infer a causality that is not proven and in fact is contraindicated by the absence of fracturing chemicals in the water wells.
Industry proponents on the other hand make statements such as “hydraulic fracturing has never contaminated ground water”. Lisa Jackson of the EPA testified recently under oath “I’m not aware of any proven case where the fracking process itself has affected water, although there are investigations ongoing.” In precise terms this may be right in that fractures have not propagated into ground water. Take the case of a well associated with fracturing operations that leaks gas but not liquid. One could argue that the poor construction would simply not have occurred but for the desire to fracture the shale reservoir. So an opponent would take those very data and say “hydraulically fractured wells contaminate ground water”, while the proponent could say “hydraulic fracturing did not contaminate ground water”. Neither would be wrong. It is the public that will be confused with this license taken with the language.
Rhetoric aside, proper stewardship of our resources and the environment is possible. Some possible measures are listed here. Permits must be given only to oil companies with good track records, thus maximizing the chances of diligence in well construction. Water wells proximal to intended operations (Jackson suggests 2,000 feet, I believe) be tested prior to drilling at the cost of the operator. Logs be required to to assure cement integrity. At a minimum the Cement Bond Log; this famously was not run on the Macondo well that blew up in the Gulf. Routine testing of the water wells, with a prompt attempt to seal the well, if leaking. This occurrence should also prompt a severe penalty. All of this and adherence to sound drilling and completion practice will ensure the sustainable production of a valuable resource.
NATURAL GAS VERSUS COAL: DUELING REPORTS
September 28, 2011 § 1 Comment
Until recently, natural gas was seen indisputably as a cleaner alternative to coal. Robert Howarth at Cornell University changed all that, at first abortively in 2009, when his study was demonstrably flawed. His revised report, which now includes the contribution of fugitive methane in coal mining, has been published. A hailstorm of criticism notwithstanding, some of the issues beg debate. A more recent study appears to be in support as well. In contrast is the report by the Worldwatch Institute, conducted in collaboration with Deutsche Bank which unequivocally concludes the superiority of natural gas, nevertheless recommends attention to fugitive emissions. 
So what is the public to make of all of this? They are right to assume that science is deterministic at least in the broad swaths of the argument in question. So there is no dispute that when combusted, natural gas produces about 50% less carbon dioxide than coal in producing the same amount of electricity. Where the dueling reports diverge is in the area of fugitive emissions: these are releases of methane during the operations involved in producing and transporting the fuels. There is also no dispute that methane is about 25 times more potent than carbon dioxide in its global warming proclivity.
The bulk of the debate surrounding the Howarth work has been around the time scale for the analysis. This is because, although methane is much more potent, it turns out that this potency dissipates much faster than in the case of carbon dioxide. So, one gets a different result when the effects are studied for twenty years as for a hundred years. The latter has been the accepted standard. But Howarth and others make an argument for using the shorter time span, which turns out to disfavor methane. Of note is the fact that when carbon sequestration in deep saline aquifers is considered, the yardstick they are held to is well in excess of a hundred years. In other words the sequestered gas has to be guaranteed to not leak over that period.
In the case of coal, the emissions comprise methane found in association with the coal. For centuries this has been a known hazard of coal mining, both from the standpoint of poisonous atmosphere for miners and from the possibility of explosions in confined areas of the mines. In the past, canaries were famously used as indicators of methane. If they died you got out in a hurry; a sort of go no-go device. Some of those still awake through this discourse no doubt are skeptical in that you know you can smell a gas leak in your kitchen. Well, it turns out methane has no odor, but the producers deliberately introduce one for precisely the intended purpose of olfactory detection of leaks.
Natural gas production and distribution can leak in two principal areas. One is in transportation. The system of pipelines and associated valve assemblies at various points can leak after aging induced malfunctions. But this can be addressed through maintenance mechanisms. The main source of fugitive emissions is the natural gas produced prior to the existence of a pipeline to move it. This is in the early days of the prospect. Even in areas riddled with pipelines, a spur line to the new rig in question does not exist at the outset. Current custom is to not invest in that until the reservoir is proven commercially viable. The initial gas produced during the discovery process has nowhere to go. It is often released. Hence the problem. Now, it could be flared, which is the process of simply burning it on the end of a pipe. This would dramatically reduce the problem since the released pollutant would be carbon dioxide, not methane. But one imagines this approach is not taken probably because flaring draws singular attention to the enterprise. This gas produced in the very early days of the well is the problem.
The public may well ask why something useful is not done with the gas. The answer lies in part in the short duration of the production. It cannot economically warrant any sort of capture and use. But if such a technology were to be developed, the potential would be significant.
A final note: fugitive methane emissions from livestock exceed that from oil and gas operations. This results from the fact that ruminants such as cows produce methane as a normal consequence of their digestive process: they belch methane. An outbreak of vegetarianism would help the environment!